
Eyecare
Latest News
Latest Videos

Podcasts
CME Content
More News

Falls among older adults with visual impairment are strongly linked to modifiable home hazards—especially broken flooring and tripping risks—while fall risk remains relatively low in hazard-free homes.
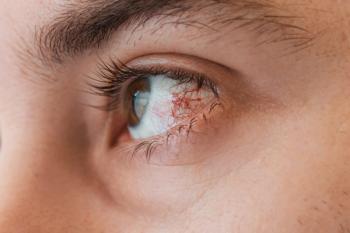
Ophthalmologists and oncologists created a new ocular adverse event grading scale for oncology trials that addresses ambiguities and provides clear dose modification recommendations.
Kamuvudines are a new class of drugs that are derived from the anti-HIV nucleoside reverse transcriptase inhibitors. They are designed to inhibit the inflammasome, part of the innate immune system that is triggered in geographic atrophy.

Eylea HD offers two dosing options for patients with retinal vein occlusion: 8-week and 4-week dosing intervals.

Lesion growth was reduced by 40.5% in patients receiving Izervay in an open-label study. A real-world study confirms safety in more than 10,000 patients.

Although dry eye disease (DED) affects millions of Americans, there is a lack of effective treatment options and discontinuation rates are high, according to an abstract presented at AMCP Nexus 2025.

Researchers say ophthalmologists and optometrists were motivated, at least in part, to use the sutureless grafts for dry eye disease by the profit margin they make on devices.
Changes in eye blood vessels signal early risk for Alzheimer’s disease, offering a noninvasive biomarker for early detection and intervention.
Increasing the cholesterol-lowering protein ApoM in mice improved retinal health and may be the key to slowing age-related macular degeneration in its earliest stages.

The UC Davis team is studying a less invasive way of delivering gene therapy to the eye: injecting the gene therapy into the suprachoroidal space, a tiny layer between the white of the eye and the blood vessels that feed the retina.

Wykoff discussed treating geographic atrophy in patients with wet age-related macular degeneration and his presentation of the results of the COAST trial that was halted earlier this year.

The American Society of Retina Specialists’ History of Retina is an initiative that highlights the milestones and the pioneers in ophthalmology and retinal disease treatment.
The vision research community rallies to protect the National Eye Institute from funding cuts, emphasizing its vital role in preventing blindness and advancing treatments.

The Vision Center at Children’s Hospital Los Angeles is developing a protocol for a phase 1 clinical trial for the first gene therapy for boys with blue cone monochromacy, which impairs color vision and severely affects visual function.

In early research, OCU410ST, a gene therapy for Stargardt disease, demonstrated slowed lesion growth by 48% at the 12-month follow-up in evaluable treated eyes.
A class of drugs, MDM2 inhibitors, being studied as cancer therapeutics may accidentally damage the protective barriers in the brain and eyes, finds new research in mice.

GLP-1 drugs such as Ozempic and Mounjaro outperformed metformin, statins and insulin in lowering rates of age-related macular degeneration and primary open-angle glaucoma.

Alcon, a global eye care company known for products such as Systane and GenTeal Tears, plans to launch Tryptyr in the U.S. market in the third quarter of 2025. International rollouts are expected to follow.

A case analysis provides a new understanding of the ocular side effects of Blenrep, an antibody drug conjugate that is being reviewed by the FDA as a combination treatment for patients with multiple myeloma. The goal date is July 23, 2025.
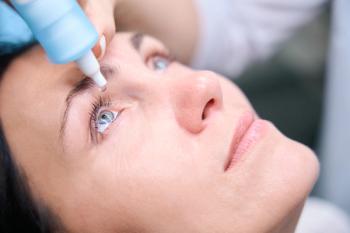
The annual dilated retinal exam, a unique tool which uses drops to dilate the pupil, is not just about eye health. It offers a comprehensive view of a member's overall well-being, potentially identifying serious health issues at an early stage.
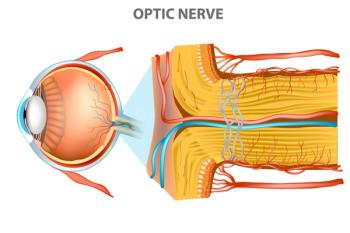
The active ingredient in Ozempic and Wegovy is linked to nonarteritic anterior ischemic optic neuropathy (NAION), the second most common form of optic neuropathy. This is apparently the first study to link semaglutide to the eye condition.

Lack of standardization has obscured complication rates in clinical trials, say a team of researchers. In results reported in the journal Ophthalmology earlier month, they list 67 ocular complications that experts agreed upon using a Delphi process,
Researchers say their findings of increased prevalence argue for screening for diabetic retinopathy in young adults.
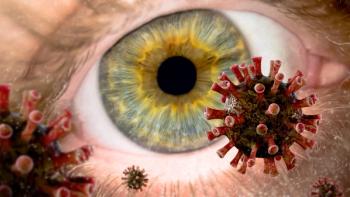
In a study in mice, researchers found that the COVID-19 virus can infect the retina even when the virus doesn’t enter through the eyes.




























