
In total, CVS Caremark has removed 26 products and added 31 products effective Jan. 1, 2023.

In total, CVS Caremark has removed 26 products and added 31 products effective Jan. 1, 2023.

The submission is expected to be complete by the end of the year, and if approved, could be available in early 2024.

Ashley Jenne, managing director, strategy and business development at Evernorth, discusses the evolution of the PBM’s digital health formulary.

The FDA uses real-world data to show how Boostrix prevents infections in infants. The agency also approves a label expansion for Oxlumo, a new administration method for Trogarzo, and accepts an sBLA for Takhzyro. In COVID-19 news, Eiger won’t submit EUA for COVID-19 treatment. Additionally, an OIG reports finds many accelerated approvals have delayed confirmatory trials.

This is the first study to use an artificial intelligence algorithm to break down visual field loss in new-onset glaucoma cases among U.S.-based population groups.

EmpiRx Health CEO Karthik Ganesh discusses how the PBM was able to decrease mental health drug spending at a time when utilization is increasing.

Medicare and Medicaid spent more than $18 billion on drugs without verified clinical benefit.
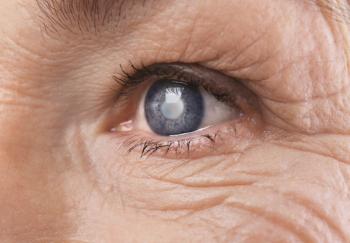
Older patients who had cataract surgery had almost a 30% lower risk of developing dementia compared with those who did not, a recent analysis finds.

Humana’s 2023 Medicare pharmacy benefits updates include $0 copays, simplified access to prescriptions and Veteran’s focused-plans that add pharmacy benefits.

If approved, valoctocogene roxaparvovec would be the first commercially-available gene therapy in the United States for the treatment of severe hemophilia A.

Researchers at the National Eye Institute have designed a gene therapy approach that could help prevent blindness in children with Leber congenital amaurosis, a rare form of blindness.

The FDA has approved several new therapies this week, including Amylyx’s oral treatment for ALS, a fourth Avastin biosimilar, a new glaucoma treatment and another indication for Duxipent. The agency also accepted an NDA for therapy for rare immunodeficiency disease.

Now with the brand name Relyvrio, the new therapy should be available within four to six weeks. It will have a wholesale acquisition cost of $158,000 per year.

Jing Yang from IQVIA discusses how national payers are using formulary exclusions for oncology therapeutics.

Carl Black, pharmacist-in-charge at Optum Rx, talks about how the PBM has decreased dispensed opioids by 62% since 2018.

But researchers found that only a small percentage of plans use available real-world evidence studies and economic evaluations in their coverage policies.

Committee members felt the data on overall survival for patients treated with Copiktra was difficult to interpret, and the therapy was associated with a higher risk of serious side effects and deaths compared with Kesimpta.

The FDA approved a tumor agnostic treatment for cancer, a lower dose MRI contrast agent, the first generic of Tazorac gel, and a gene therapy for a rare disease. Advisory committees vote down poziotinib for NSCLC and Pepaxto for multiple myeloma and give positive vote for microbiotic-based C. diff therapy. The agency has accepted sNDA for Tukysa for HER2 positive colorectal cancer.
While some patients with relapsed or refractory multiple myeloma saw a benefit in a confirmatory trial, the main issue concerning the committee members was a high rate of death in the study.
RBX2660 — now with the brand name Rebyota — is a potential first-in-class microbiota-based live biotherapeutic that aims to reduce recurrent C. difficile infection.
Several concerns factored into the decision, including a low overall response rate and high rate of adverse events, a dose that was not optimized and a confirmatory trial that will not likely see results until 2026.
Hospitalization, disability, having private high-deductible, Medicare Advantage, or no coverage were risk factors associated with medical debt.

The FDA has approved new therapies for psoriasis and kidney dysfunction. The agency has also scheduled an advisory committee meeting for nonprescription birth control and accepted applications for longer-acting aripiprazole, Rett syndrome drug, and an enzyme replacement therapy. Additionally, Clovis has submits supplemental applications for Rubraca.

The applications are based on progression-free survival data even though the FDA has recommended that the company wait for more mature overall survival data.

Terlivaz is the first approved therapy to treat hepatorenal syndrome, a rare form of kidney disfunction related to advanced liver disease.

Timothy Mizak and Nicole Bulochnik of Abarca talked about how can PBMs help payers manage costs through a hybrid network model that leverages value-based and other innovative drug contracts at the Pharmacy Benefit Management Institute (PBMI) annual meeting in Orlando.

Jeffrey A. Shaman, Ph.D., chief science officer at Coriell Life Sciences, talked about how pharmacogenomics can inform medication management.

Jeffrey A. Shaman, Ph.D., chief science officer at Coriell Life Sciences, talked about how pharmacogenomics can inform medication management.

Greg Warren, partner and consulting actuary at Axene Health Partners, discussed the many solutions that have been put forth to address the prescription drug challenges that could bring value to healthcare.
If approved, Perrigo’s Opill could be the first-ever over-the-counter birth control pill. The advisory committee meeting is scheduled for Nov. 18, 2022.