New Trial to Investigate Osteoporosis Drug for Type 1 Diabetes
The study explores whether Prolia/Xgeva (denosumab), a widely used therapy for osteoporosis and bone tumors, can regenerate beta cells in patients with early type 1 diabetes.
The City of Hope Medical Center has begun a
Amgen’s denosumab is approved as Prolia to treat osteoporosis and as Xgeva to treat bone tumors. Additionally, four sets of biosimilars have been approved by the FDA, the most recent being Fresenius’s Conexxence/Bomyntra. Additional approved biosimilars include Celltrion’s Stoboclo/Osenvelt, Samsung’s Ospomyv/Xbryk and Sandoz’s Jubbonti/Wyost.
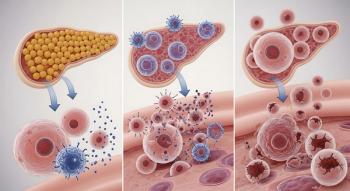
Type 1 diabetes is an autoimmune disease that affects about 2 million Americans, about 5% to 10% of all diabetes cases, according to the Centers for Disease Control and Prevention (CDC). In type 1 diabetes, the immune system’s T cells destroy the insulin-producing beta cells. For patients with type 1 diabetes, the standard of care treatment has been insulin replacement.
City of Hope’s study, which is now open, will explore whether denosumab can protect and regenerate beta cells, which produce insulin to regulate blood sugar, in type 1 diabetes patients. In the early stages of type 1 diabetes, some beta cells are still alive and functioning.
If successful, denosumab could provide protection from the autoimmune attack for any source of beta cells. Researchers believe the same mechanism that halts bone degradation might also stop the autoimmune attack on islet cells.
In the bone, denosumab works by inhibiting a protein called RANKL (Receptor Activator of Nuclear Factor Kapp-B Ligand), blocking its interaction with the receptor RANK, thereby reducing bone damage.
“We have found that the same RANKL/RANK pathway can destroy beta cells,”
Vasavada said that previous studies in their lab have shown that denosumab can inhibit the RANKL pathway and improve beta cell health by protecting, increasing the number and/or improving the function of beta cells. By protecting and preserving beta cell function, the medication could slow down the progression of type 1 diabetes and improve blood sugar control.
City of Hope is currently enrolling patients for the study, said
Primary funding for this clinical trial comes from Breakthrough T1D, formerly JDRF, the leading global type 1 diabetes research and advocacy organization.
The multicenter clinical trial will evaluate the safety and efficacy of denosumab for improving beta cell function and blood sugar control among patients with early type 1 diabetes, who continue to make some insulin.
Patients are being randomized with a 2:1 ratio to either denosumab or placebo, with 30 people enrolled to receive a denosumab 60 milligrams injection given under the skin every three months for a total of four injections. The placebo arm will enroll 15 people and be administered with normal saline placebo. Patients will be followed for 12 months for adverse events and tested for changes in beta cell function and blood sugar control.
Two additional clinical sites — the University of Alabama at Birmingham and Indiana University — are also participating in this trial.
Newsletter
Get the latest industry news, event updates, and more from Managed healthcare Executive.